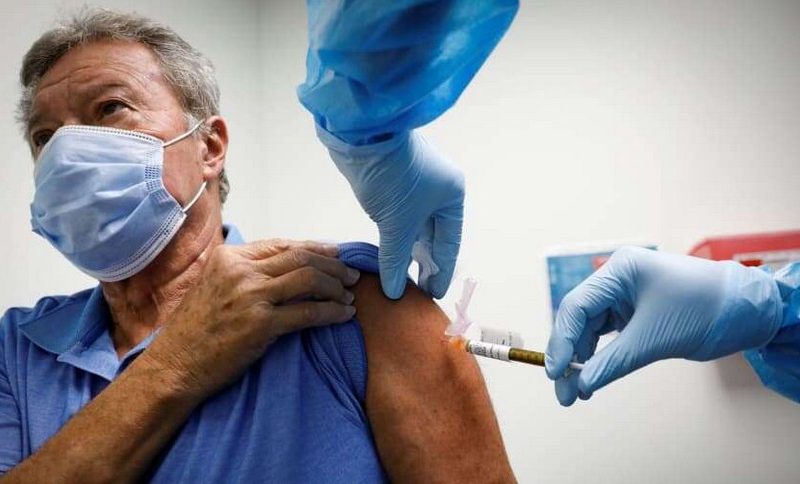
The coronavirus vaccines developed by Pzifer-BioTech would be distributed in the UK within the next few days, in line with the recent approval granted by regulators in the country, KMBC reports. The implementation of treatment is a significant step towards ending the coronavirus pandemic in the country, officials said.
In the United States, the UK, and across Europe, cases of coronavirus infections have been increasing in recent days. Public health officials are predicting a surge in cases as the holiday season begins. Even with the arrival of a vaccine, officials said it would take months before a significant portion of the populace is vaccinated, thereby heralding the herd immunity needed to eradicate the pandemic.
The Medicines and Healthcare Products Regulatory Agency, the agency tasked with approving drugs for use in the UK, gave the approval for the emergency use of the vaccine after a series of clinical trials and rigorous reviews. The vaccine had proved to be 95% effective and offered a lifeline for the older population affected by the virus, and also those with huge complications from infections.
The US FDA, countries in Europe and regulators in Canada are all in line towards approving the vaccine by Pfizer. A vaccine by Moderna and AstraZeneca also stands the chance to be approved before long, CNET writes.
According to the UK Health Secretary Matt Hancock, the UK will receive about 800,000 doses of the Pfizer vaccine within the next few days. Before the end of the year, the UK expects delivery of about millions of the vaccine. He confirmed that people will get vaccinated as soon as the vaccines arrive. To be protected, individuals need two doses of the vaccines. The two doses would be taken in the space of three weeks.
Sean Marett, a top official with BioNTech, said the drugmaker would supply 570 million doses of the vaccines globally in 2021, with options to supply 600 million more, according to deals it had signed with countries all over the world. However, in China, a local company, Fosun Pharma will deal with the distribution.
In the UK, the plan is to first vaccinate those at risk of dying from the disease and medical personnel. Other groups will follow immediately as more supplies become available.
Dr. June Raine, head of the MHPRA said that the public should be assured of the safety of the vaccine, considering the number of review processes and clinical trials the drug underwent. He said there is the need to assure the public of the efficacy and safety of the vaccine, which would be critical if the vaccination of a significant portion of the public is going to be a success.
However, logistics surrounding the distribution of the vaccine is another problem as the vaccines must be kept under certain stipulated temperature specifications until they get to individuals. The vaccine by Pzifer-BioNTech must be stored at -700C (-940F). However, the company said its shipping containers would take care of that challenge.
There are side effects to taking the vaccine except for temporary pain and flu-like reactions, according to the companies. However, there’s no word yet on how long the vaccine would last, and its effects on pregnant women.
Source: kmbc.com